Research

 Multisensory Integration
Multisensory Integration
The brain assembles different sensory stimuli into a coherent picture through fast and flexible communication mechanisms, such as neural synchronization within and between brain regions. Multisensory integration is a fundamental property of brain circuits, dynamically interacting with motor processing, cognition and attentional mechanisms. Our recent work uses computational modeling to investigate how the brain determines stimulus coherence and integrates multisensory information.



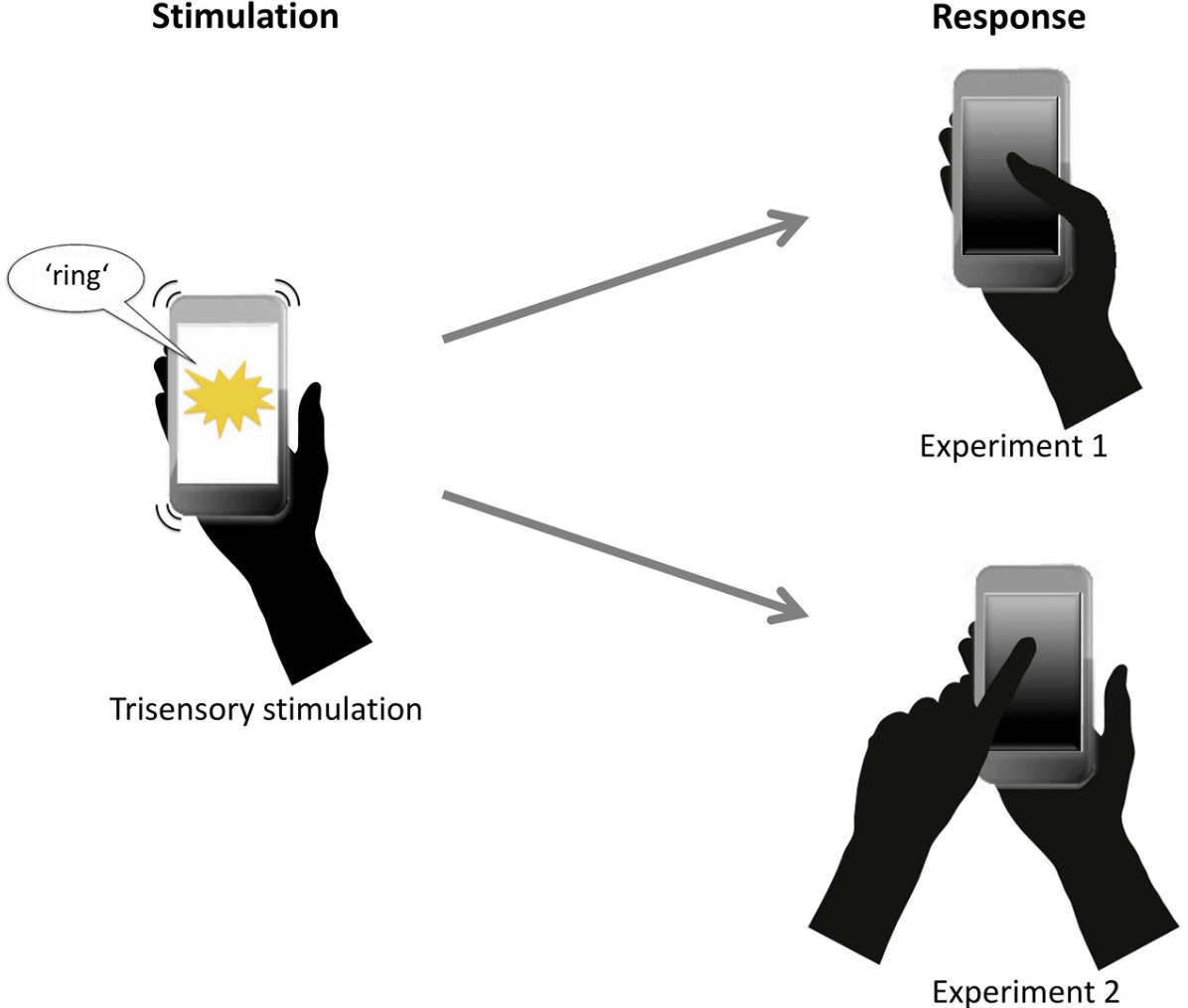

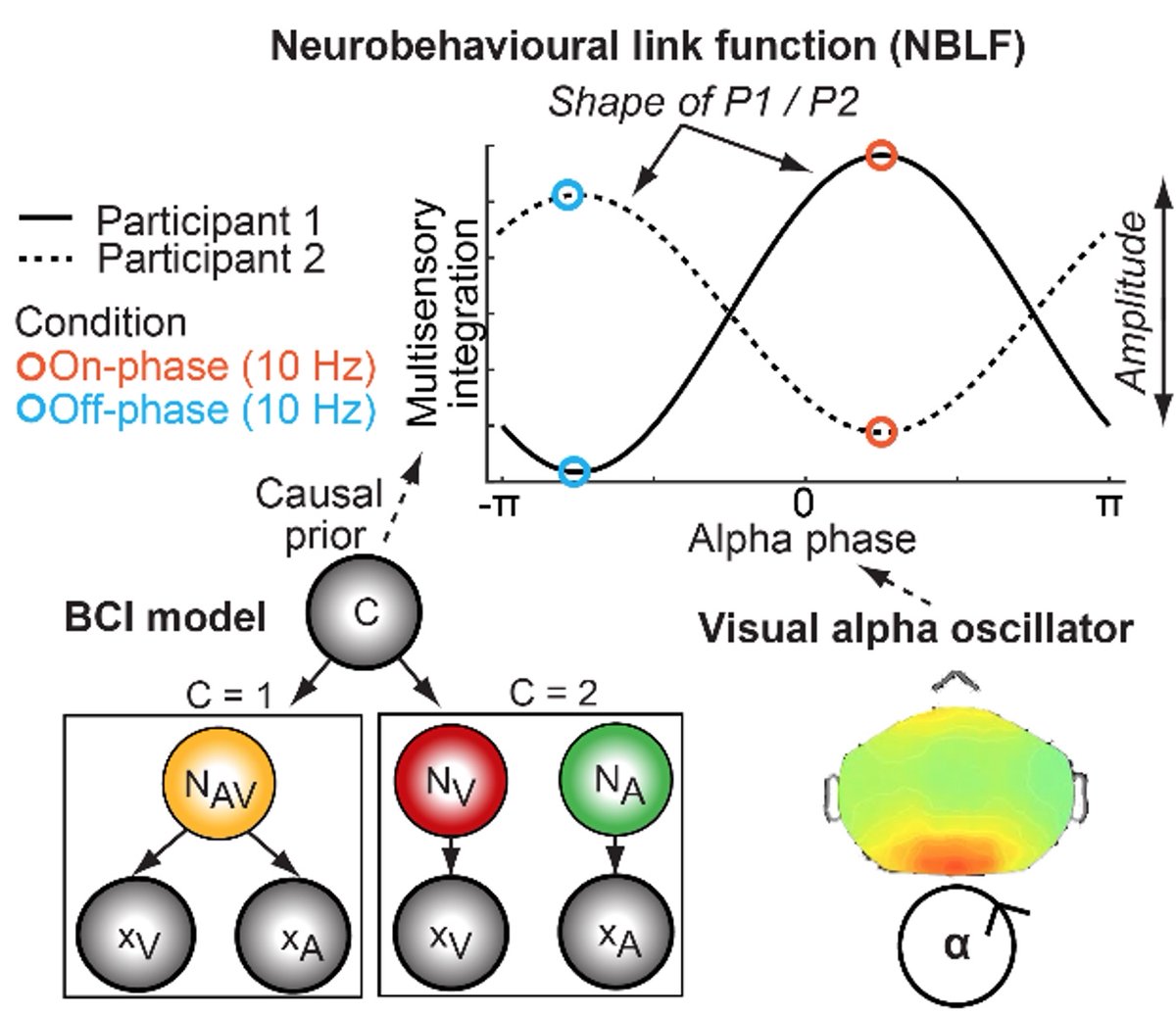
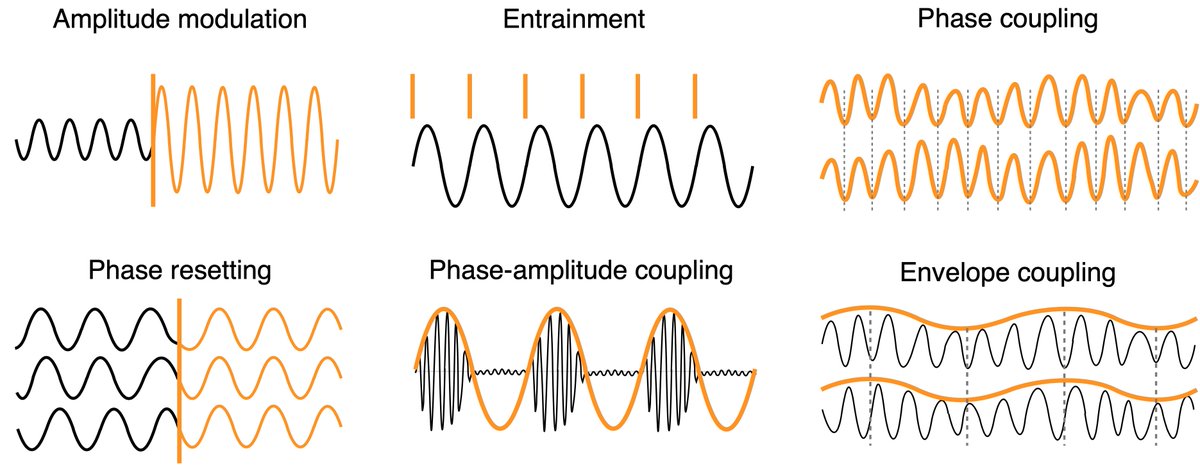


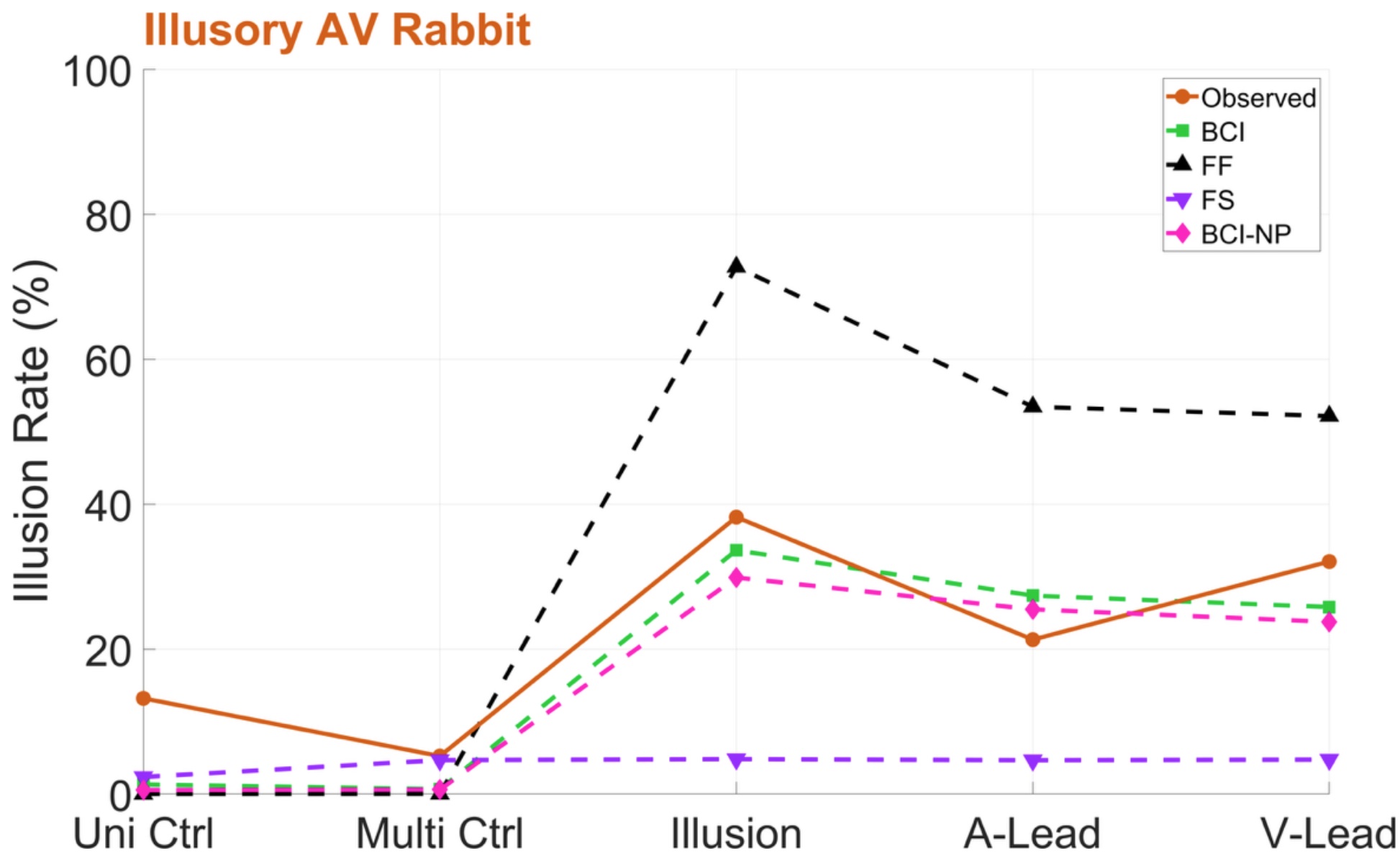



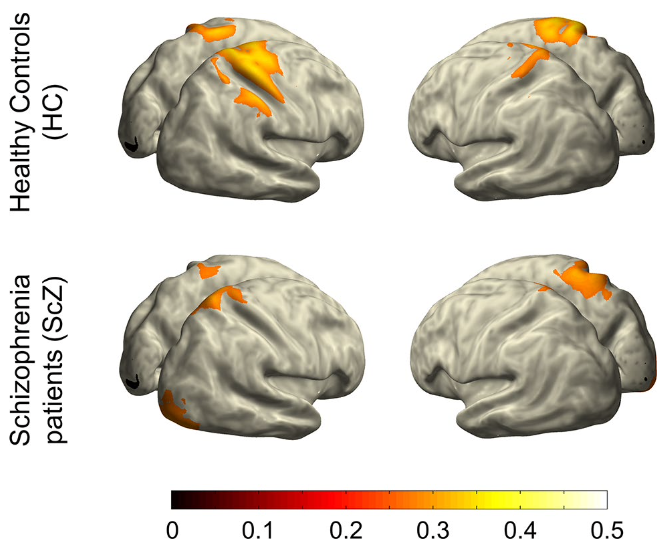
 Neural signatures of schizophrenia
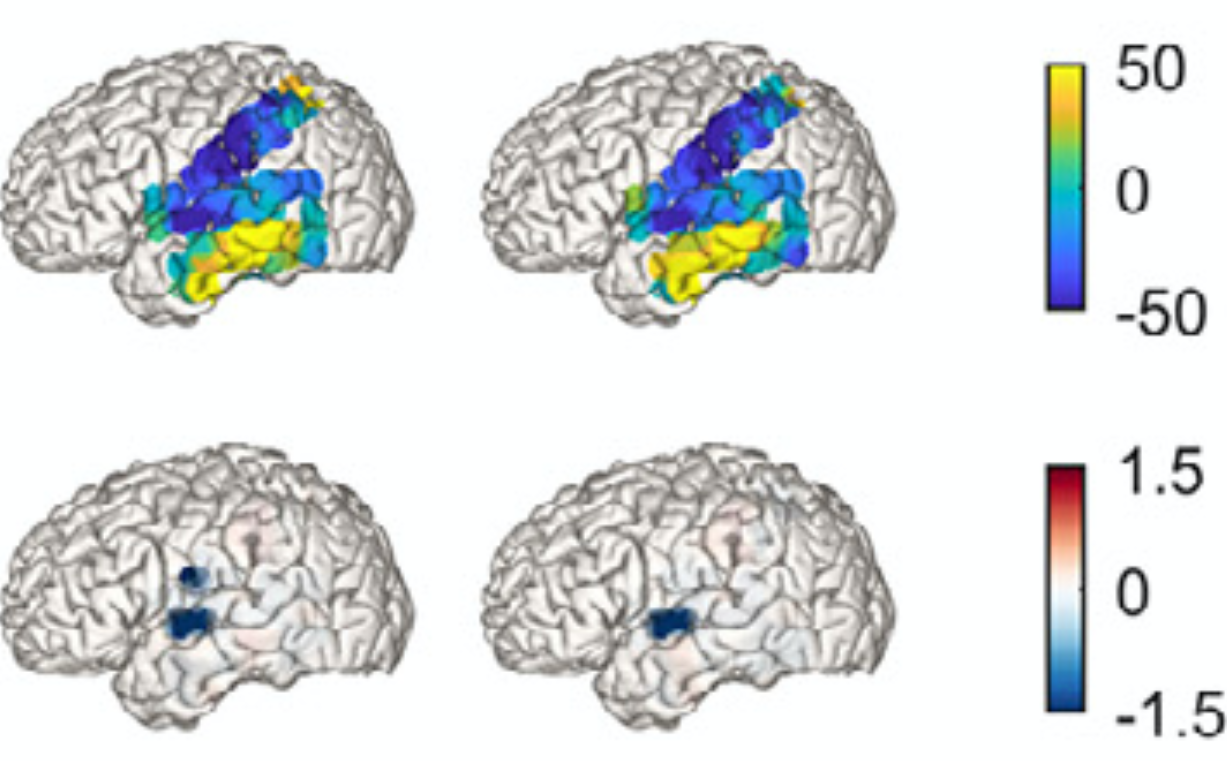
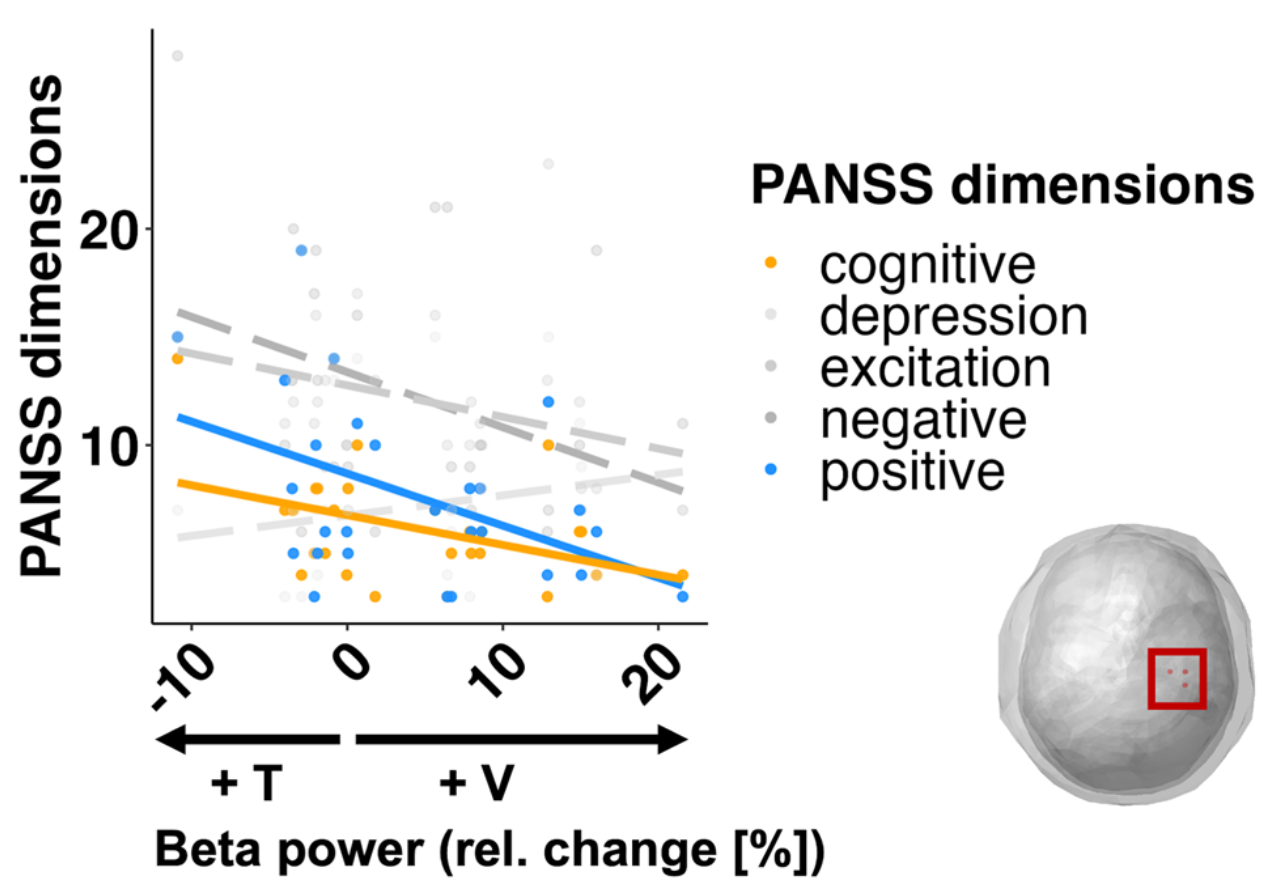
Neural signatures of schizophrenia
Beyond positive and negative symptoms, schizophrenia patients experience subtle perceptual and cognitive deficits, likely driven by altered neural synchronization. Surprisingly, multisensory processing shows only minor impairments and can even compensate for attentional deficits. We are currently investigating whether working memory deficits are generalizable or subfunction-specific.



 Crossmodal processing of pain
Crossmodal processing of pain
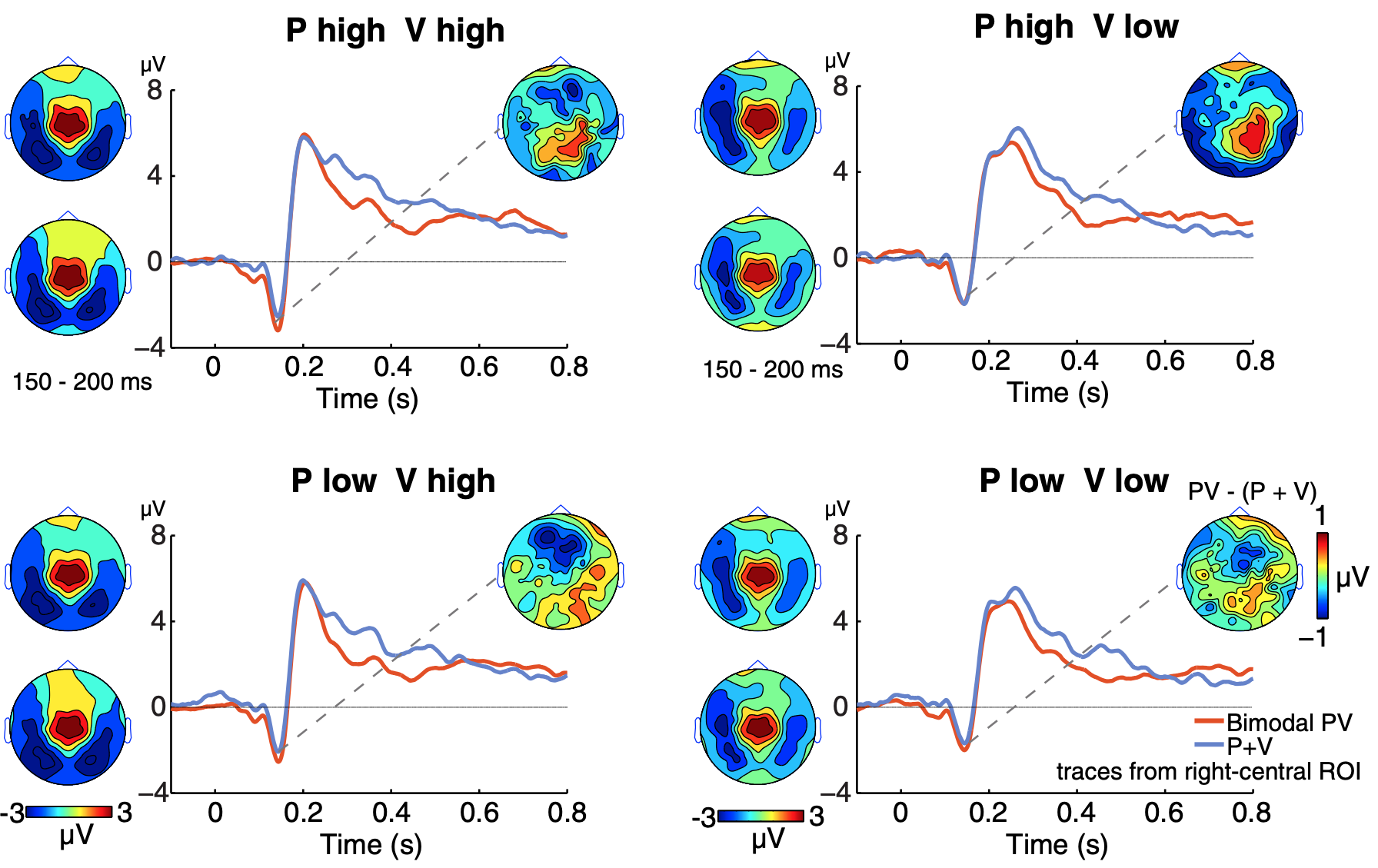
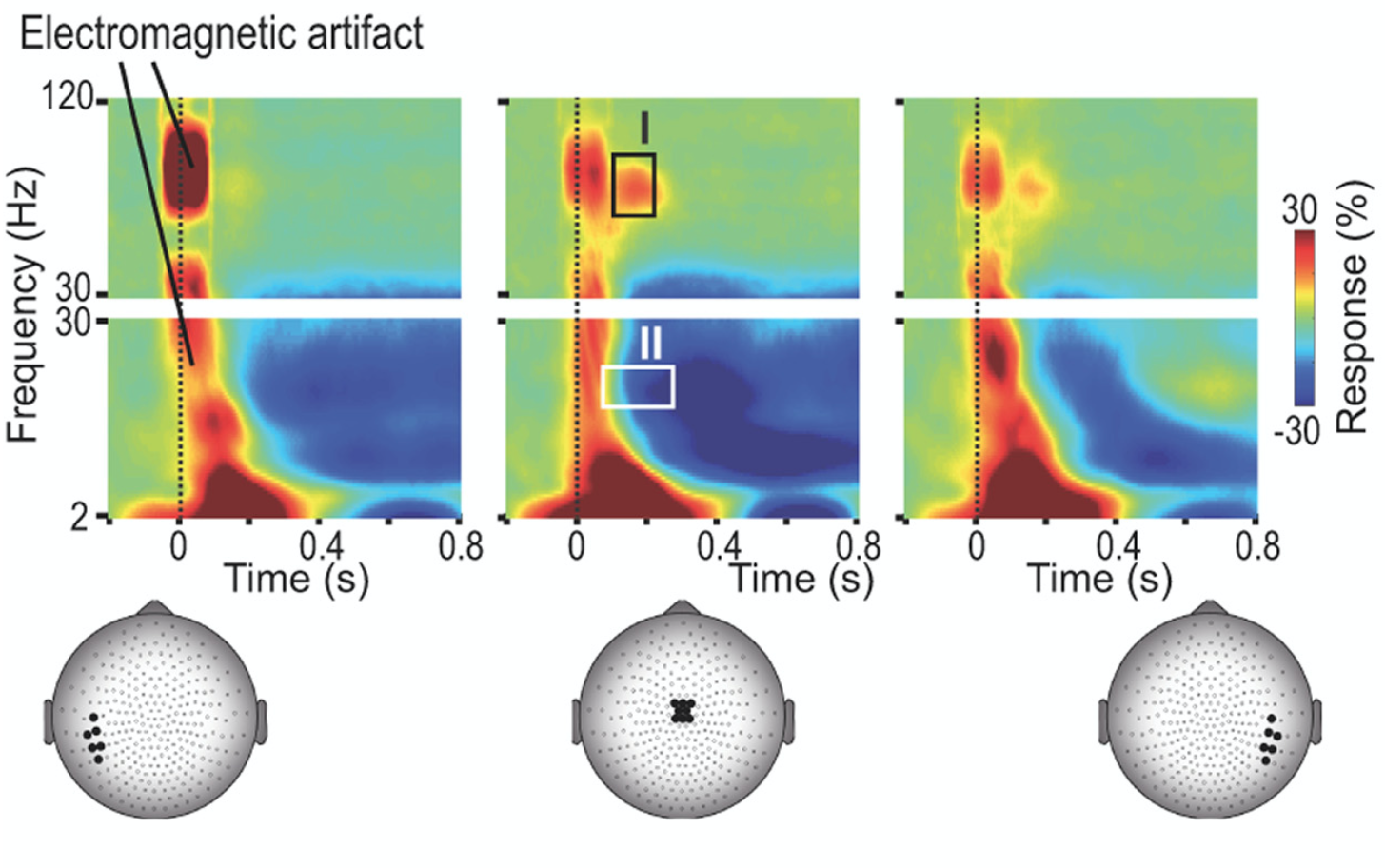
Our research examines how sensory input from other modalities affects acute pain processing. Such crossmodal stimuli can either reduce pain by distraction or enhance it by increasing salience. We have demonstrated this bidirectional modulation of pain perception and processing. Additionally, we hypothesize that chronic pain distorts body representation in the brain, with implications for developing virtual reality interventions for chronic pain treatment.




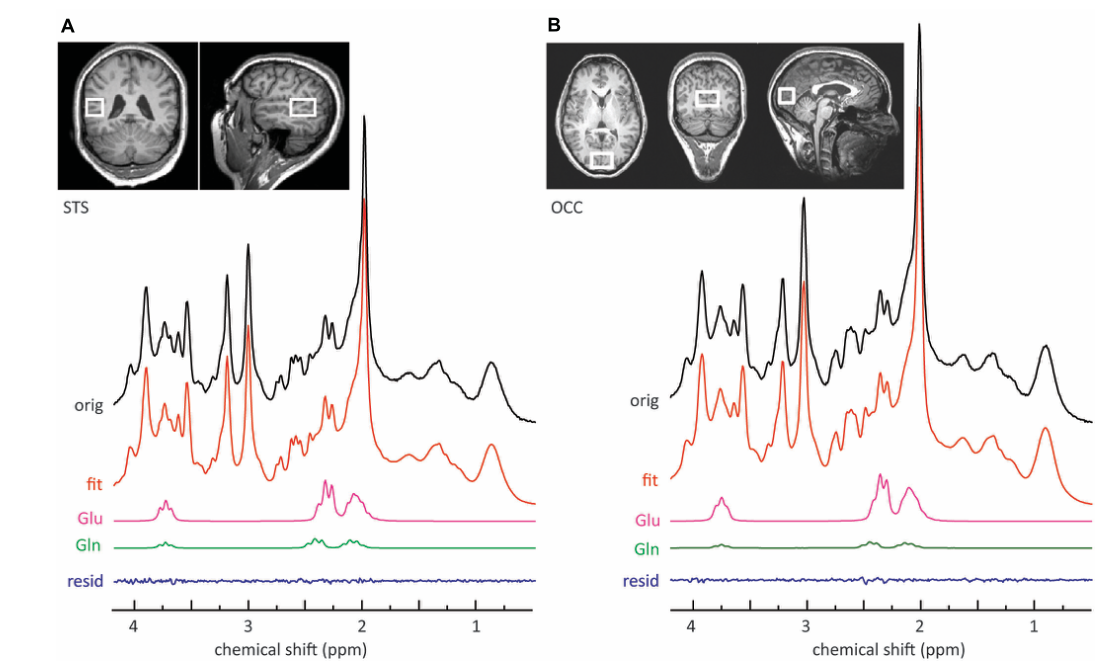
Other topics
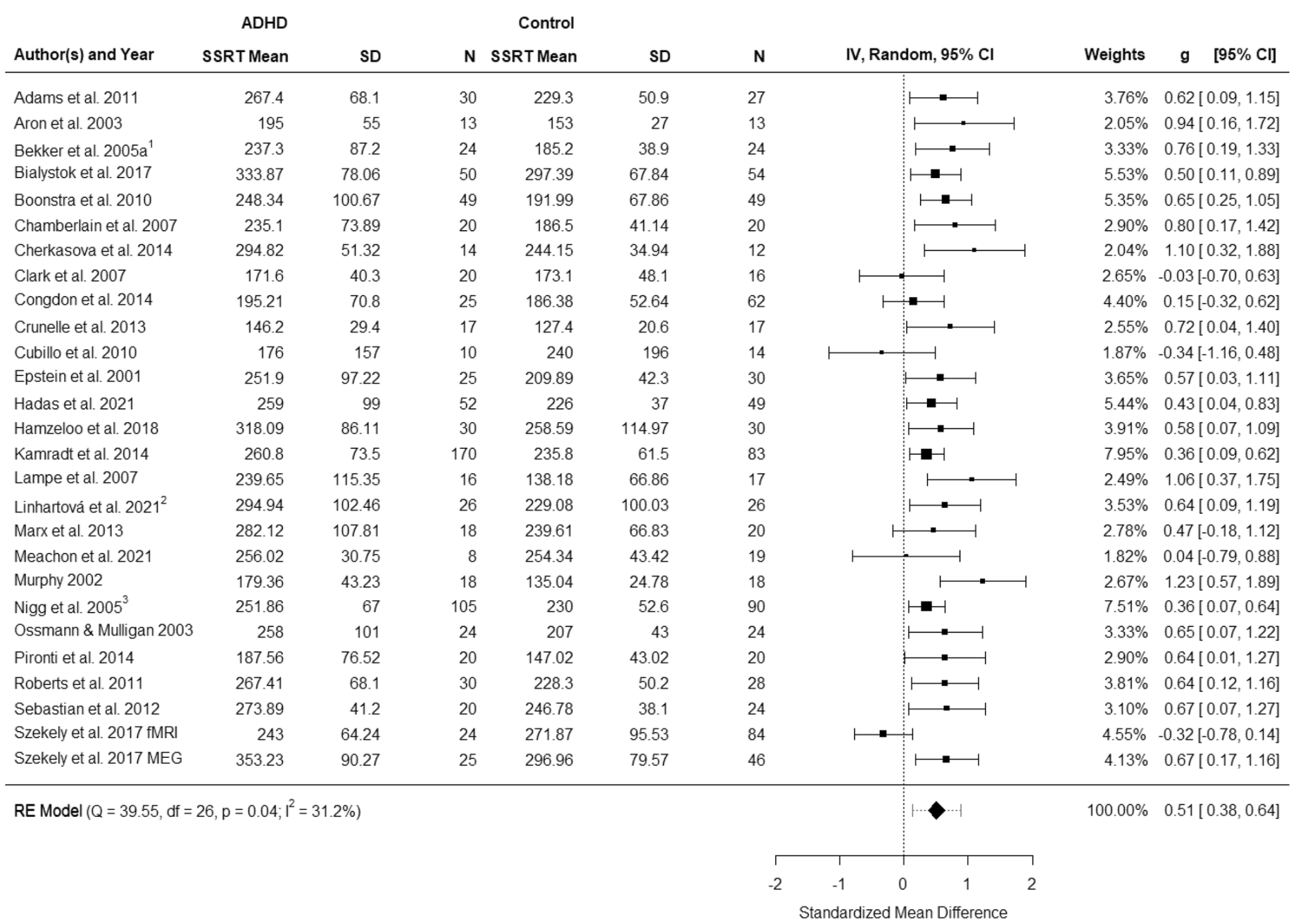
We have conducted projects on diverse topics including adult ADHD, generalized anxiety disorder, cochlear implants, genetics, and MR spectroscopy. Currently, we are investigating memory processing in PTSD, applying knowledge of dynamic neural memory processes to understanding memory dysfunction, which is a core feature where traumatic memories become fragmented and intrusive.



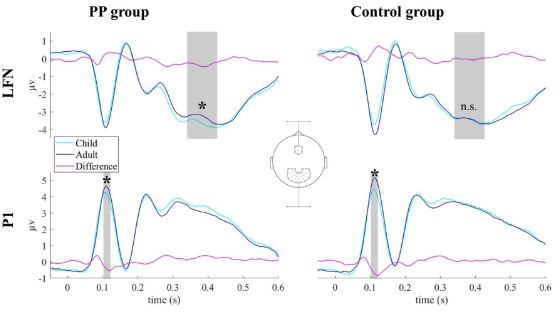
processing of child face stimuli in prepubescent children.Speer et al., 2022, Eur Child Adol Psychiatr